Awareness Workshop

21/5/26
ΝΕΟ FSSC 22000 Version 7: Μετάβαση και Νέες Απαιτήσεις
Ημερίδα
SPECIALISED WEBINAR
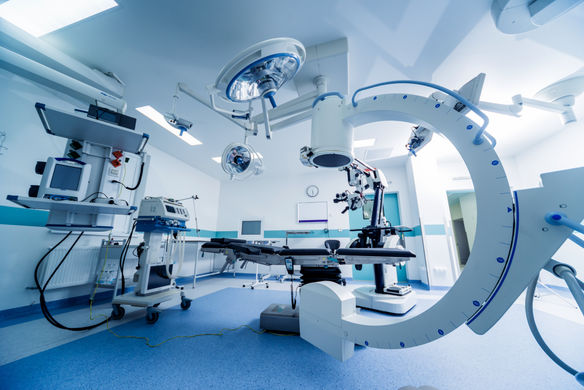
13/5/26
MDR 2017/745 TECHNICAL DOCUMENTATION REQUIREMENTS FOR THE ORTHOPEDIC INDUSTRY
Ημερίδα
Awareness Workshop

20/4/26
TÜV NORD Ελλάδας | Awareness Workshop #7: ΝΕΟ ISO 14001:2026: Τι αλλάζει και πώς επηρεάζει τον Οργανισμό σας
Ημερίδα
Awareness Workshop

4/3/26
TÜV NORD Ελλάδας | Awareness Workshop #6: Μία προσεκτική ανάγνωση στο νέο πλαίσιο για την εκπαίδευση των χειριστών τροφίμων (ΥΑ 110581/2025, ΦΕΚ 2161 Β/06-05-2025)
Ημερίδα
Awareness Workshop

4/2/26
TÜV NORD Ελλάδας | Awareness Workshop #5: Η Συνέντευξη ως Στιγμή Απόφασης
Ημερίδα
Awareness Workshop

8/12/25
TÜV NORD Ελλάδας | Awareness Workshop #4: Ο νέος Νόμος για τη «Δίκαιη Εργασία για όλους».
Ημερίδα
Awareness Workshop

31/10/25
TÜV NORD Ελλάδας | Awareness Workshop #3: Το ISO 9001:2015 αναθεωρείται – Προετοιμαστείτε για το ISO 9001:2026
Ημερίδα
Awareness Workshop

15/10/25
TÜV NORD Ελλάδας | Awareness Workshop #2: Πώς να αναρτήσετε την Έκθεση CO₂ στην Πλατφόρμα ΟΦΥΠΕΚΑ
Read the full article